SOHA AMREEN
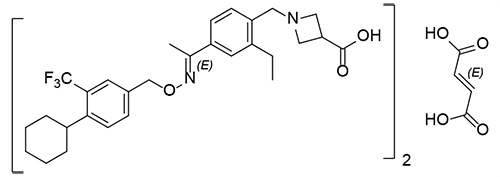
- Siponimod is an oral, second generation sphingosine-1-phosphate receptor modulator. S1P receptor modulators can inhibit the egress and recirculation of lymphocytes from lymph nodes, a therapeutic strategy for treating certain autoimmune diseases.
INDICATION: Siponimod is the first and only treatment for patients with active secondary progressive multiple sclerosis (SPMS)
MECHANISM OF ACTION: Siponimod causes a dose-dependent decrease of the peripheral blood lymphocyte count within 6 hours of the first dose, caused by the reversible accumulation of lymphocytes in lymphoid tissues, due to lack of lymphocyte release. This results in a decrease in the inflammation that is involved in multiple sclerosis.
PHARMACOKINETICS
ABSORPTION: The time (Tmax) to attain maximum plasma concentrations (Cmax) after oral administration of immediate-release oral doses of siponimod was found to be approximately 4 hours (with a range 3 – 8 hours).
DISTRIBUTION: Siponimod distributes to body tissues with an average volume of distribution of 124 L Protein binding of siponimod is higher than 99.9% in healthy patients as well as hepatic and renal impaired patients
METABOLISM: Siponimod is extensively metabolized, mainly by CYP2C9 enzyme (79.3%), and subsequently by CYP3A4 enzyme (18.5%)
ELIMINATION: Siponimod is eliminated from the systemic circulation mainly due to metabolism, and subsequent biliary/fecal excretion.
half-life is approximately 30 hours
DOSAGE: The recommendation is for approval of Siponimod tablets 0.25 mg, 1 mg and 2 mg, for treatment of secondary progressive multiple sclerosis (SPMS), an advanced-stage of autoimmune disease of multiple sclerosis.
EVIDENCES: It is found that 64.8% of patients experienced sustained clinical disease stability after 12 months of siponimod therapy. The review also noted that 21.4% of stable patients improved, while 31.5% experienced EDSS progression